Sourcing Surgical Instruments from Sialkot: A Quality Guide
BLOGS
3/29/2026

Sialkot, Pakistan, is the undisputed world capital for the manufacturing of hand-held surgical instruments. Over 70% of the world's supply of forceps, scalpels, and scissors originate from this single industrial hub.
For US medical supply companies, buying directly from Sialkot offers massive profit margins. However, because the US healthcare market is strictly regulated, maintaining flawless quality control is not optional—it is a legal requirement.
Here is a simple guide on how US buyers can safely source medical instruments from Sialkot while protecting their business from liability.
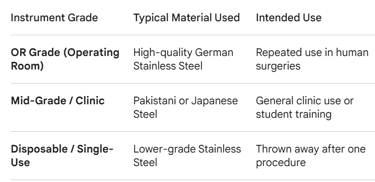
The 3 Tiers of Surgical Instruments
Before requesting quotes, you must understand that Sialkot factories produce three different grades of instruments. You need to know exactly what you are buying:

4 Quality Control Rules for US Buyers
1. Demand FDA Registration and ISO Certificates
The US Food and Drug Administration (FDA) treats surgical instruments as medical devices.
Never work with a factory that is not FDA-registered.
Ask for proof of ISO 13485 certification (the specific international quality standard for medical devices).
2. Verify the Steel Quality (The "Boil Test")
The most common issue with cheap instruments is rusting after sterilization. To prevent this, professional buyers demand a "Boil Test."
A sample instrument is boiled in distilled water for 30 minutes.
If any rust or discoloration appears, the steel alloy used is defective or low-grade.
3. Hire a Local Third-Party Inspector
Do not rely on the factory to check its own work. Hire an independent inspection agency in Sialkot (like SGS or QIMA) to go into the factory and check your batch before it is packed.
They will randomly pull 10% of the order to check for sharp edges, proper joint movement, and correct laser marking.
4. Require Proper Passivation
Passivation is a chemical process that removes free iron from the surface of the stainless steel, creating a shield that prevents rusting. Ask your supplier for a certificate proving that the batch went through a proper chemical passivation bath.
Checklist for US Medical Importers
[ ] Verify the supplier's FDA registration number.
[ ] Request a physical sample of the exact instrument grade you need.
[ ] Run a boil test on the provided sample.
[ ] Hire an on-ground inspection company for final random testing.
[ ] Ensure all instruments have the "Country of Origin" (Pakistan) laser-etched on the metal.
© 2025. All rights reserved.
Contacts:
info@karobardirectory.com
